- Services
Services
Inoviem provides a full range of services from drug discovery to clinical development by leveraging its platforms and direct access to human pathological specimens.
All the platforms and services are label-free with no modification of the original molecules.Unravel disease relevant Mode of Action (MoA) of your molecule directly on human tissues.
Identify and validate targets in your study model or in Human.
Thanks to our ex-vivo pharmacology expertise we identify the best disease and patients subgroups.
Using patients’ samples and PIMS® technology, we identify those individuals who can benefit best from your compound as soon as the early stage of preclinical studies.
Compounds of interest can be profiled for their drugability based on their mode of action and efficacy.
Translational pharmacology empowers your ability to identify biomarkers and advance clinical developments.
Advanced SPR and proteomics drive precise analysis of biomolecular interactions and protein landscapes.
Meticulous collection and biobanking of human samples fuel robust, translational research.
- Platforms
Platforms
Inoviem provides breakthrough protein technologies for each phase of the drug development process.
One of the major advantages of our label-free technologies used under physiological conditions and in human tissue – NPOT® and PIMS® – is that the test compound retains its original structure, as well as the exact same molecular structure that would be used in therapyPIMS®
Designed to study molecular interactions and predict therapeutic responses in biomedical research
Biomarkers discovery
Methodology for the proteomic profiling of human individuals in the context of translational evaluation of biomarkers and clinical development of new drug candidates.
- Knowledge centre
- About us
About us
Inoviem Scientific is a Bioanalytical R&D company established in 2011 in Strasburg – France.

Our mission
Our mission is clear: to get the right treatment to the right patients within the right therapeutic window.
- Contact us
Indications
- Malarial parasites infections
- Rheumatoid arthritis
- Discoid or systemic lupus
- Erythematosus lupus
Side effects
- Headache
- Nausea or indigestion
- Diarrhoea
- Skin rashes, bleaching of the hair
Pharmacological effect
Exact mechanism of action is unknown
- erythrocytic dev. of plasmodia
- pH of the acid vesicles
- phospholipid metabolism
- production of rheumatoid factors
- HCQ is accumulated in white blood cells
Toxicity
Over long period of medication or exposition at high doses, HCQ increases the risk of eye damage, including vision changes and induces rare but irreversible retinopathies (3-5%).1
- Lupus market: 5 million people throughout the world2
- Rheumatoid arthritis market: 350 million people worldwide3
- ON target identification on human lupus PBMCs
- OFF target identification on mice retina
- Design of a new molecule that led to a patent application: WO2016193503
- Efficacy & Safety validation of the new molecule
- In vitro: Functional/Activity assay, SPR
- In vivo: 2 mouse models (MRL/lpr & C57Bl/6)
- Early ADME: Pharmacokinetics
- Study completed in 9 months
Peripheral Blood Mononuclear Cell (PBMC) lysates from 20 lupus patients under treatment and retinas from 5 mice were used to perform NPOT® experiments in combination with HCQ, in order to identify HCQ’s ON and OFF targets that could explain its anti-inflammatory activity and retinal toxicity.
This experiment allowed to identify Myeloperoxydase (MPO) as the ON target with occurrence rate of 70%, whereas alphaB-crystallin (αB-C) was identified as the OFF target with occurrence rate of 80%.
MPO has an antimicrobial activity4 and is a key enzyme for neutrophil extracellular trap (NET)5 that explains very well HCQ’s anti-inflammatory properties (figure 1).
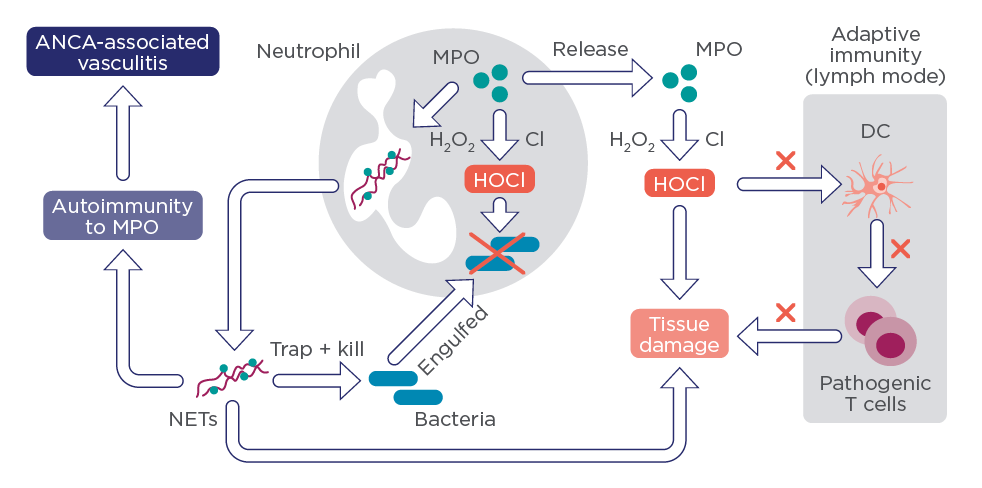
Figure 1: The role of MPO on Neutrophil-Mediated Regulation of Immunity (Dragana Odobasic et al. Journal of Immunology Research 2016; ID 2349816)
αB-C is however known to be expressed in many parts of the retina6 and vitreous fluid. Recent studies have shown that αB-C promotes tumor angiogenesis7 and retinal neovascularization8.
The biophysical interaction of HCQ with its targets has been validated by SPR (Biacore 3000®): Kd of 7,1.10-7M on MPO and 2,2.10-8M on its OFF target and functional assay by quantifying HCQ’s influence on the MPO activity (figure 2).
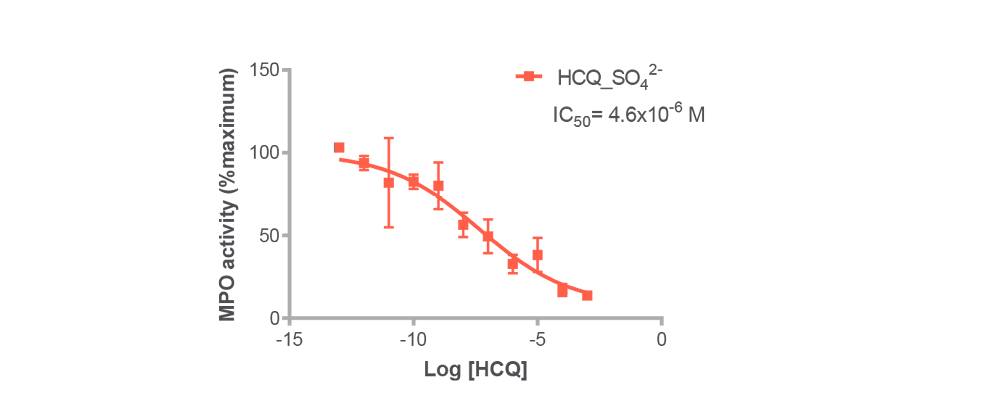
Figure 2: Influence of HCQ concentration on MPO activity
With the aim of loosing the OFF binding interaction, a serie of analogues has been designed and synthesized (in collaboration with the University of Strasbourg, France) based on the chemical structures of MPO and αB-C. The candidate INS-072 has been selected for its enhanced anti-inflammatory properties (INS- 072 EC50 = 6,9.10-9M vs HCQ EC50 = 1,9.10-7M), for its greater efficacy against dermatitis appearance in the MRL/lpr mice model that is known to spontaneously develop Lupus symptoms (figure 3A) and finally for its reduced emergence of retinopathy (figure 3B) in the C57Bl/6 mice model that is sensible to visual damage.
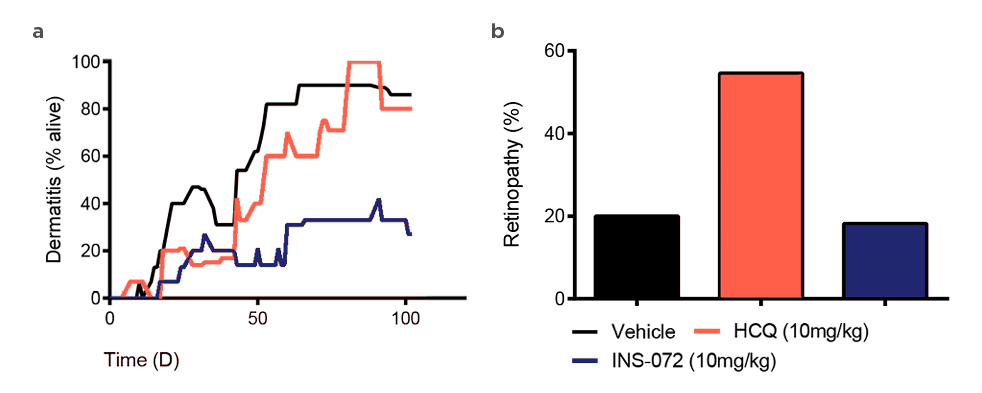
Figure 3:
A) Representation of the pourcentage of dermatitis appearance over time (in days)
B) Analysis of the cornea Integrity (in pourcentage of retinopathy)
CONCLUSION
Thanks to NPOT®, Inoviem Scientific was capable of identifying and validating, not only HCQ’s ON & OFF targets (Myeloperoxydase and alphaB-crystallin) that explain its unknown mode of action, but also a new drug candidate with a better efficacy and reduced side effect observed on mouse retinas in systemic autoimmune disease.
REFERENCES
- 1. Latasiewicz et al. Eye (Lond) 2017; 31(6): 972–976
- 2. www.ressources.lupus.org
- 3. www.medicinenet.com
- 4. https://www.ncbi.nlm.nih.gov/gene?cmd=retrieve&list_uids=4353.
- 5. Metzler et al. Blood 2011; 117(3):953-9
- 6. Reddy et al. Current molecular medicine 2015; 15:47-61
- 7. RDimberg et al. Blood 2008; 111:2015-23
- 8. Xu et al. Investigative ophtalmology & visual science 2015; 56:816-26
